Draw The Best Lewis Structure For Ccl+13. What Is The Formal Charge On The C?
Chapter vii. Chemical Bonding and Molecular Geometry
7.3 Lewis Symbols and Structures
Learning Objectives
By the terminate of this department, you will be able to:
- Write Lewis symbols for neutral atoms and ions
- Draw Lewis structures depicting the bonding in simple molecules
Thus far in this chapter, we have discussed the various types of bonds that grade between atoms and/or ions. In all cases, these bonds involve the sharing or transfer of valence beat out electrons betwixt atoms. In this department, nosotros volition explore the typical method for depicting valence vanquish electrons and chemical bonds, namely Lewis symbols and Lewis structures.
Lewis Symbols
Nosotros use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. A Lewis symbol consists of an elemental symbol surrounded by 1 dot for each of its valence electrons:
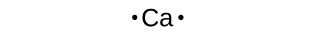
Figure ane shows the Lewis symbols for the elements of the 3rd flow of the periodic tabular array.

Lewis symbols can besides be used to illustrate the formation of cations from atoms, as shown here for sodium and calcium:

Too, they tin can be used to show the formation of anions from atoms, as shown here for chlorine and sulfur:

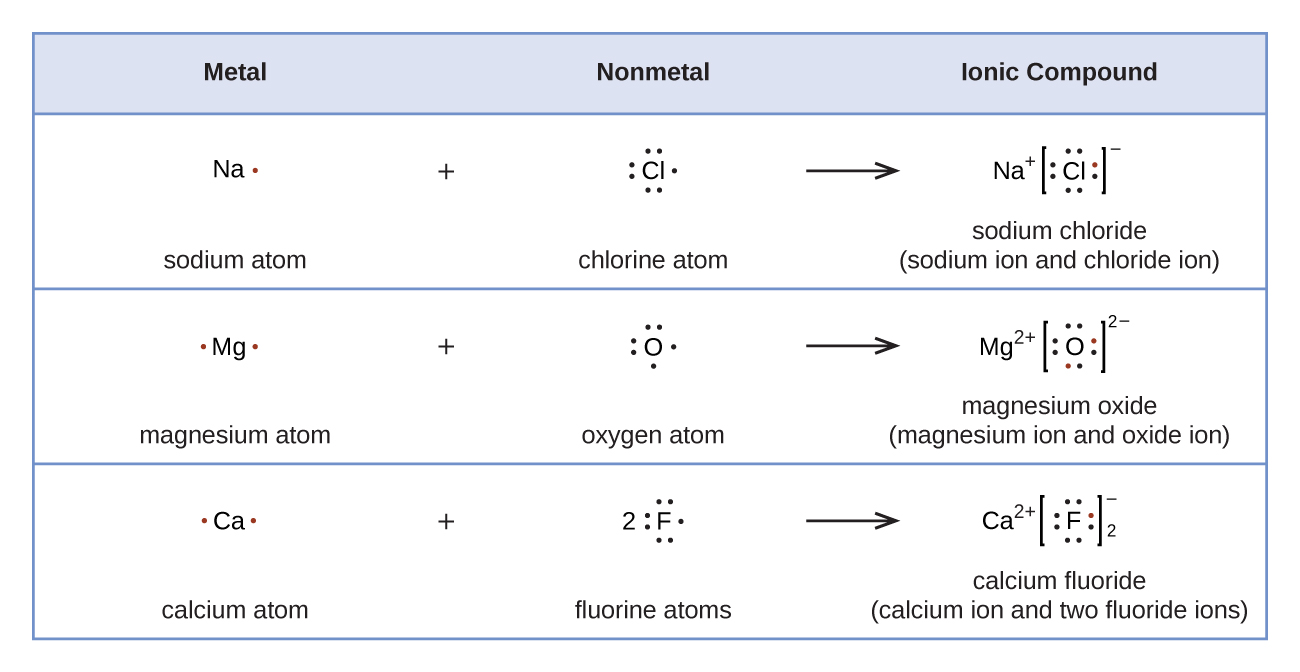
Figure 2 demonstrates the apply of Lewis symbols to show the transfer of electrons during the formation of ionic compounds.
Lewis Structures
Nosotros besides use Lewis symbols to bespeak the formation of covalent bonds, which are shown in Lewis structures, drawings that describe the bonding in molecules and polyatomic ions. For example, when 2 chlorine atoms form a chlorine molecule, they share ane pair of electrons:
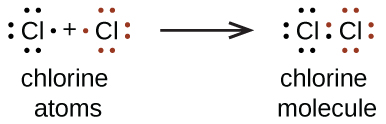
The Lewis structure indicates that each Cl atom has three pairs of electrons that are non used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). A nuance (or line) is sometimes used to point a shared pair of electrons:

A single shared pair of electrons is called a single bail. Each Cl atom interacts with eight valence electrons: the six in the lonely pairs and the 2 in the single bond.
The Octet Rule
The other halogen molecules (F2, Brtwo, Iii, and Attwo) form bonds like those in the chlorine molecule: one single bond betwixt atoms and three lone pairs of electrons per atom. This allows each halogen atom to have a noble gas electron configuration. The trend of master group atoms to form enough bonds to obtain 8 valence electrons is known as the octet rule.
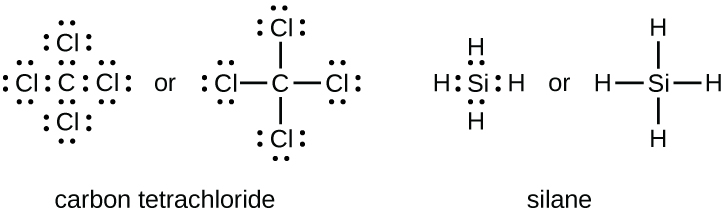
The number of bonds that an atom can class tin can often be predicted from the number of electrons needed to reach an octet (eight valence electrons); this is especially true of the nonmetals of the 2nd menses of the periodic table (C, North, O, and F). For example, each atom of a group 14 element has 4 electrons in its outermost shell and therefore requires four more than electrons to attain an octet. These four electrons can be gained past forming four covalent bonds, as illustrated here for carbon in CCl4 (carbon tetrachloride) and silicon in SiH4 (silane). Because hydrogen merely needs two electrons to make full its valence shell, it is an exception to the octet rule. The transition elements and inner transition elements also do non follow the octet rule:
Grouping xv elements such every bit nitrogen accept five valence electrons in the diminutive Lewis symbol: one alone pair and 3 unpaired electrons. To obtain an octet, these atoms form three covalent bonds, as in NH3 (ammonia). Oxygen and other atoms in group 16 obtain an octet by forming ii covalent bonds:
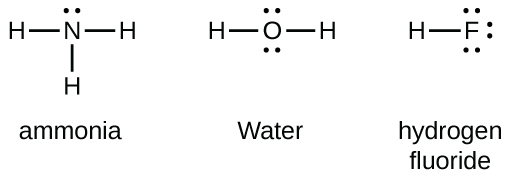
Double and Triple Bonds
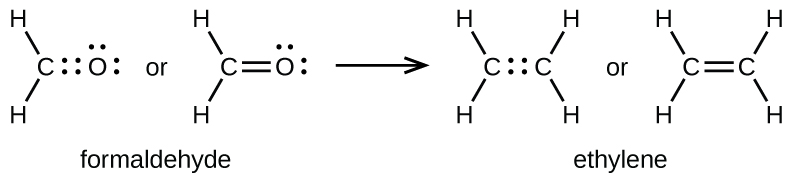
As previously mentioned, when a pair of atoms shares 1 pair of electrons, nosotros call this a single bail. Nevertheless, a pair of atoms may need to share more than 1 pair of electrons in order to achieve the requisite octet. A double bail forms when two pairs of electrons are shared between a pair of atoms, equally between the carbon and oxygen atoms in CHtwoO (formaldehyde) and between the ii carbon atoms in CiiH4 (ethylene):
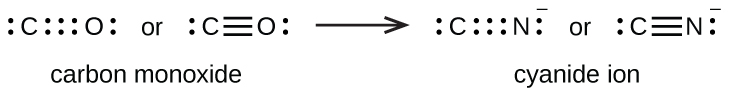
A triple bail forms when iii electron pairs are shared by a pair of atoms, as in carbon monoxide (CO) and the cyanide ion (CN–):
Writing Lewis Structures with the Octet Rule
For very simple molecules and molecular ions, we can write the Lewis structures by merely pairing upwards the unpaired electrons on the constituent atoms. See these examples:
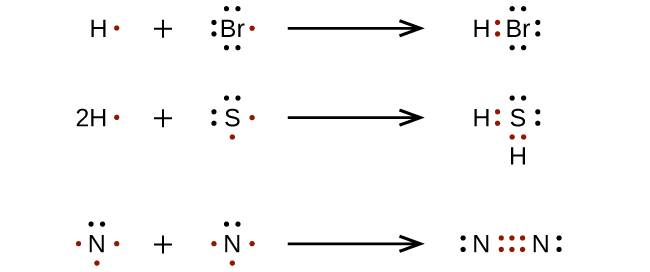
For more complicated molecules and molecular ions, information technology is helpful to follow the step-by-footstep procedure outlined here:
- Determine the total number of valence (outer vanquish) electrons. For cations, decrease 1 electron for each positive charge. For anions, add one electron for each negative charge.
- Describe a skeleton structure of the molecule or ion, arranging the atoms around a central atom. (Generally, the least electronegative element should be placed in the center.) Connect each atom to the central atom with a single bail (one electron pair).
- Distribute the remaining electrons as lone pairs on the terminal atoms (except hydrogen), completing an octet around each atom.
- Identify all remaining electrons on the central atom.
- Rearrange the electrons of the outer atoms to make multiple bonds with the central cantlet in club to obtain octets wherever possible.
Let u.s.a. decide the Lewis structures of SiH4, CHOii−, NO+, and OF2 every bit examples in following this procedure:
- Determine the total number of valence (outer shell) electrons in the molecule or ion.
- For a molecule, we add the number of valence electrons on each atom in the molecule:
[latex]\begin{assortment}{r r l} \text{SiH}_4 & & \\[1em] & \text{Si: four valence electrons/atom} \times 1 \;\text{cantlet} & = iv \\[1em] \rule[-0.5ex]{21em}{0.1ex}\hspace{-21em} + & \text{H: 1 valence electron/atom} \times 4 \;\text{atoms} & = 4 \\[1em] & & = 8 \;\text{valence electrons} \end{array}[/latex]
- For a negative ion, such as CHOtwo −, we add together the number of valence electrons on the atoms to the number of negative charges on the ion (i electron is gained for each single negative charge):
[latex]\begin{array}{r r l} {\text{CHO}_2}^{-} & & \\[1em] & \text{C: four valence electrons/cantlet} \times one \;\text{atom} & = 4 \\[1em] & \text{H: 1 valence electron/atom} \times 1 \;\text{atom} & = 1 \\[1em] & \text{O: 6 valence electrons/atom} \times 2 \;\text{atoms} & = 12 \\[1em] \rule[-0.5ex]{21.5em}{0.1ex}\hspace{-21.5em} + & one\;\text{additional electron} & = i \\[1em] & & = 18 \;\text{valence electrons} \end{array}[/latex]
- For a positive ion, such as NO+, we add the number of valence electrons on the atoms in the ion and then subtract the number of positive charges on the ion (one electron is lost for each single positive charge) from the total number of valence electrons:
[latex]\begin{array}{r r l} \text{NO}^{+} & & \\[1em] & \text{Northward: 5 valence electrons/atom} \times ane \;\text{atom} & = 5 \\[1em] & \text{O: six valence electrons/atom} \times i \;\text{atom} & = 6 \\[1em] \rule[-0.5ex]{21em}{0.1ex}\hspace{-21em} + & -1 \;\text{electron (positive charge)} & = -1 \\[1em] & & = 10 \;\text{valence electrons} \end{array}[/latex]
- Since OFtwo is a neutral molecule, nosotros only add together the number of valence electrons:
[latex]\begin{array}{r r l} \text{OF}_{2} & & \\[1em] & \text{O: 6 valence electrons/atom} \times one \;\text{cantlet} & = 6 \\[1em] \rule[-0.5ex]{21em}{0.1ex}\hspace{-21em} + & \text{F: 7 valence electrons/atom} \times two \;\text{atoms} & = 14 \\[1em] & & = xx \;\text{valence electrons} \end{array}[/latex]
- For a molecule, we add the number of valence electrons on each atom in the molecule:
- Draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom and connecting each atom to the central atom with a single (one electron pair) bail. (Annotation that we denote ions with brackets around the structure, indicating the charge outside the brackets:)
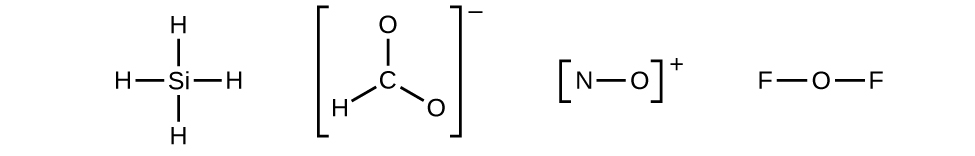 When several arrangements of atoms are possible, as for CHO2 −, we must use experimental evidence to choose the correct 1. In general, the less electronegative elements are more likely to be fundamental atoms. In CHO2 −, the less electronegative carbon cantlet occupies the central position with the oxygen and hydrogen atoms surrounding it. Other examples include P in POCl3, S in And so2, and Cl in ClO4 −. An exception is that hydrogen is most never a central atom. Equally the most electronegative element, fluorine also cannot be a central cantlet.
When several arrangements of atoms are possible, as for CHO2 −, we must use experimental evidence to choose the correct 1. In general, the less electronegative elements are more likely to be fundamental atoms. In CHO2 −, the less electronegative carbon cantlet occupies the central position with the oxygen and hydrogen atoms surrounding it. Other examples include P in POCl3, S in And so2, and Cl in ClO4 −. An exception is that hydrogen is most never a central atom. Equally the most electronegative element, fluorine also cannot be a central cantlet. - Distribute the remaining electrons as lonely pairs on the terminal atoms (except hydrogen) to consummate their valence shells with an octet of electrons.
- There are no remaining electrons on SiHfour, so it is unchanged:
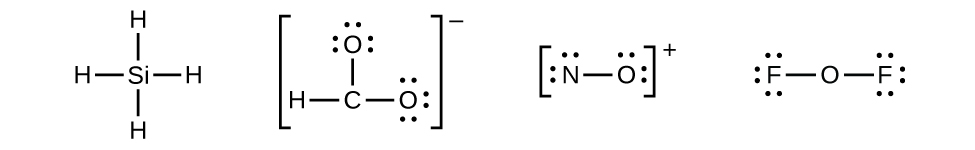
- There are no remaining electrons on SiHfour, so it is unchanged:
- Place all remaining electrons on the primal cantlet.
- For SiH4, CHO2 −, and NO+, there are no remaining electrons; we already placed all of the electrons determined in Step 1.
- For OF2, we had 16 electrons remaining in Step 3, and we placed 12, leaving iv to exist placed on the key atom:
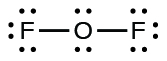
- Rearrange the electrons of the outer atoms to make multiple bonds with the fundamental cantlet in order to obtain octets wherever possible.
Instance one
Writing Lewis Structures
NASA's Cassini-Huygens mission detected a large cloud of toxic hydrogen cyanide (HCN) on Titan, one of Saturn's moons. Titan also contains ethane (H3CCH3), acetylene (HCCH), and ammonia (NHthree). What are the Lewis structures of these molecules?
Solution
- Calculate the number of valence electrons.HCN: (1 × i) + (4 × i) + (5 × 1) = 10HiiiCCH3: (ane × 3) + (2 × 4) + (1 × three) = 14HCCH: (1 × ane) + (two × 4) + (1 × 1) = 10NH3: (5 × one) + (iii × 1) = 8
- Draw a skeleton and connect the atoms with single bonds. Remember that H is never a cardinal atom:
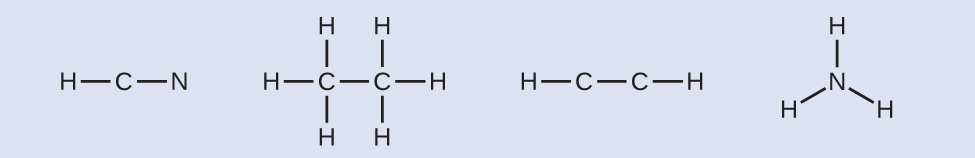
- Where needed, distribute electrons to the terminal atoms:
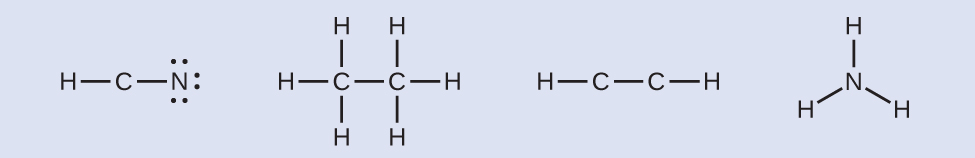 HCN: six electrons placed on NHthreeCCHthree: no electrons remainHCCH: no terminal atoms capable of accepting electrons
HCN: six electrons placed on NHthreeCCHthree: no electrons remainHCCH: no terminal atoms capable of accepting electrons NH3: no terminal atoms capable of accepting electrons
- Where needed, place remaining electrons on the central atom:
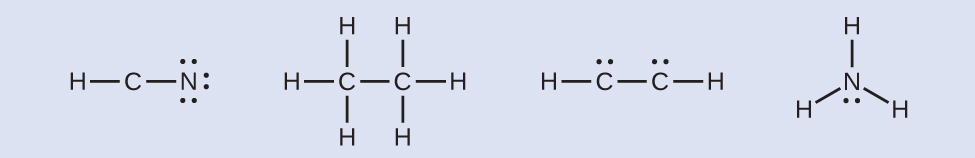 HCN: no electrons remainH3CCH3: no electrons remainHCCH: four electrons placed on carbon
HCN: no electrons remainH3CCH3: no electrons remainHCCH: four electrons placed on carbon NH3: two electrons placed on nitrogen
- Where needed, rearrange electrons to form multiple bonds in order to obtain an octet on each atom:HCN: class two more than C–N bondsH3CCH3: all atoms have the correct number of electronsHCCH: grade a triple bail between the ii carbon atomsNHiii: all atoms take the correct number of electrons
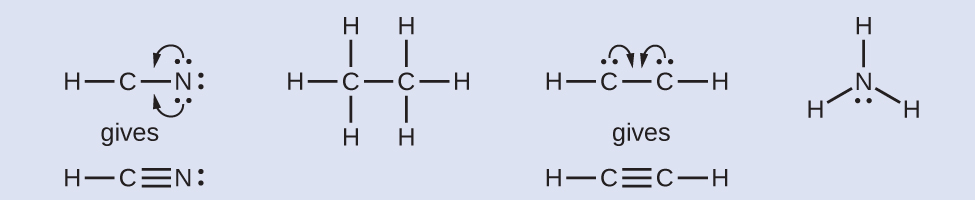
Check Your Learning
Both carbon monoxide, CO, and carbon dioxide, CO2, are products of the combustion of fossil fuels. Both of these gases besides crusade problems: CO is toxic and CO2 has been implicated in global climatic change. What are the Lewis structures of these two molecules?
Reply:

Fullerene Chemistry
Carbon soot has been known to man since prehistoric times, but it was not until fairly recently that the molecular structure of the primary component of soot was discovered. In 1996, the Nobel Prize in Chemical science was awarded to Richard Smalley (Figure 3), Robert Roll, and Harold Kroto for their work in discovering a new form of carbon, the C60 buckminsterfullerene molecule (Effigy 1 in Chapter 7 Introduction). An entire form of compounds, including spheres and tubes of various shapes, were discovered based on Cthreescore. This type of molecule, called a fullerene, shows promise in a diverseness of applications. Because of their size and shape, fullerenes can encapsulate other molecules, so they have shown potential in diverse applications from hydrogen storage to targeted drug delivery systems. They as well possess unique electronic and optical properties that have been put to good utilize in solar powered devices and chemical sensors.

Exceptions to the Octet Rule
Many covalent molecules have key atoms that do not have eight electrons in their Lewis structures. These molecules fall into 3 categories:
- Odd-electron molecules have an odd number of valence electrons, and therefore take an unpaired electron.
- Electron-deficient molecules accept a central atom that has fewer electrons than needed for a element of group 0 configuration.
- Hypervalent molecules have a central cantlet that has more electrons than needed for a noble gas configuration.
Odd-electron Molecules
Nosotros call molecules that contain an odd number of electrons free radicals. Nitric oxide, NO, is an example of an odd-electron molecule; it is produced in internal combustion engines when oxygen and nitrogen react at high temperatures.
To draw the Lewis construction for an odd-electron molecule like NO, we follow the same 5 steps nosotros would for other molecules, only with a few minor changes:
- Make up one's mind the total number of valence (outer shell) electrons. The sum of the valence electrons is 5 (from North) + half-dozen (from O) = 11. The odd number immediately tells us that nosotros have a free radical, and so we know that non every atom can have eight electrons in its valence shell.
- Depict a skeleton structure of the molecule. We can hands describe a skeleton with an North–O single bond:N–O
- Distribute the remaining electrons as alone pairs on the terminal atoms. In this case, there is no central atom, and then we distribute the electrons effectually both atoms. We give 8 electrons to the more electronegative cantlet in these situations; thus oxygen has the filled valence vanquish:
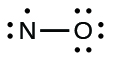
- Place all remaining electrons on the cardinal atom. Since in that location are no remaining electrons, this footstep does non apply.
- Rearrange the electrons to brand multiple bonds with the central atom in order to obtain octets wherever possible. We know that an odd-electron molecule cannot have an octet for every atom, just nosotros want to become each atom as shut to an octet as possible. In this instance, nitrogen has just five electrons effectually it. To motility closer to an octet for nitrogen, nosotros accept one of the lone pairs from oxygen and apply it to grade a NO double bond. (We cannot take some other lonely pair of electrons on oxygen and course a triple bond because nitrogen would so have nine electrons:)
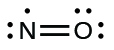
Electron-deficient Molecules
We will also run into a few molecules that contain central atoms that do non accept a filled valence crush. Generally, these are molecules with central atoms from groups ii and 12, outer atoms that are hydrogen, or other atoms that do not form multiple bonds. For example, in the Lewis structures of beryllium dihydride, BeHii, and boron trifluoride, BFthree, the beryllium and boron atoms each have merely 4 and 6 electrons, respectively. It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in BFiii, satisfying the octet dominion, but experimental evidence indicates the bail lengths are closer to that expected for B–F single bonds. This suggests the best Lewis structure has three B–F unmarried bonds and an electron deficient boron. The reactivity of the compound is also consequent with an electron scarce boron. Even so, the B–F bonds are slightly shorter than what is actually expected for B–F unmarried bonds, indicating that some double bond character is found in the bodily molecule.
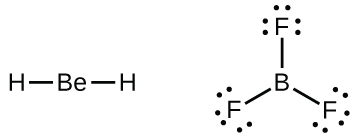
An atom similar the boron atom in BF3, which does not take eight electrons, is very reactive. It readily combines with a molecule containing an cantlet with a lone pair of electrons. For instance, NH3 reacts with BFiii because the solitary pair on nitrogen can be shared with the boron atom:
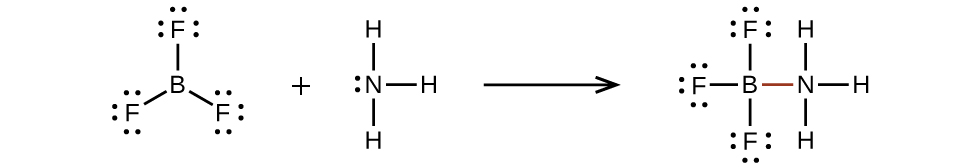
Hypervalent Molecules
Elements in the second flow of the periodic tabular array (n = two) can accommodate only eight electrons in their valence shell orbitals because they have merely 4 valence orbitals (one 2s and three 2p orbitals). Elements in the 3rd and higher periods (north ≥ 3) take more than than four valence orbitals and tin share more iv pairs of electrons with other atoms because they accept empty d orbitals in the aforementioned shell. Molecules formed from these elements are sometimes called hypervalent molecules. Figure iv shows the Lewis structures for two hypervalent molecules, PCl5 and SFhalf-dozen.
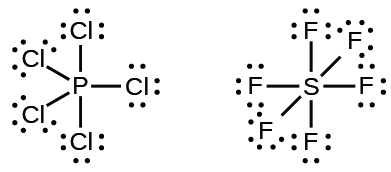
In some hypervalent molecules, such as IFfive and XeF4, some of the electrons in the outer shell of the central atom are lone pairs:

When we write the Lewis structures for these molecules, we discover that we have electrons left over later on filling the valence shells of the outer atoms with viii electrons. These boosted electrons must be assigned to the fundamental atom.
Example 2
Writing Lewis Structures: Octet Rule Violations
Xenon is a noble gas, simply it forms a number of stable compounds. We examined XeF4 before. What are the Lewis structures of XeF2 and XeF6?
Solution
Nosotros can draw the Lewis structure of whatever covalent molecule by post-obit the 6 steps discussed earlier. In this case, we can condense the last few steps, since not all of them apply.
- Summate the number of valence electrons: XeF2: 8 + (2 × 7) = 22XeFhalf-dozen: 8 + (6 × seven) = 50
- Describe a skeleton joining the atoms by single bonds. Xenon will be the central cantlet considering fluorine cannot be a central cantlet:
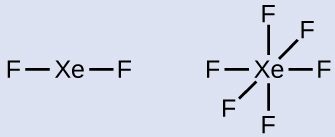
- Distribute the remaining electrons.XeFtwo: We identify three lonely pairs of electrons around each F cantlet, accounting for 12 electrons and giving each F atom viii electrons. Thus, 6 electrons (three lone pairs) remain. These lone pairs must be placed on the Xe atom. This is acceptable because Xe atoms have empty valence shell d orbitals and can adapt more than viii electrons. The Lewis structure of XeFii shows two bonding pairs and 3 lonely pairs of electrons effectually the Xe atom:
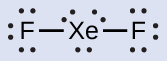 XeF6: We identify iii solitary pairs of electrons around each F atom, bookkeeping for 36 electrons. Ii electrons remain, and this lone pair is placed on the Xe atom:
XeF6: We identify iii solitary pairs of electrons around each F atom, bookkeeping for 36 electrons. Ii electrons remain, and this lone pair is placed on the Xe atom: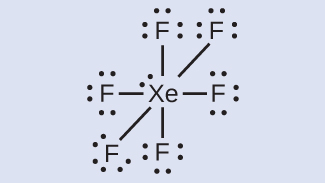
Check Your Learning
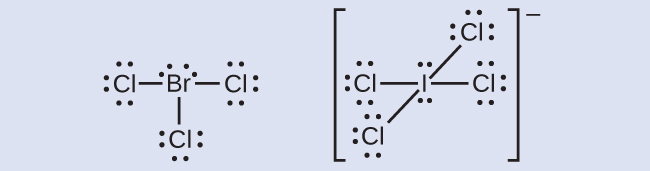
The halogens course a grade of compounds chosen the interhalogens, in which halogen atoms covalently bond to each other. Write the Lewis structures for the interhalogens BrCliii and ICl4 −.
Respond:
Key Concepts and Summary
Valence electronic structures can be visualized by cartoon Lewis symbols (for atoms and monatomic ions) and Lewis structures (for molecules and polyatomic ions). Lone pairs, unpaired electrons, and single, double, or triple bonds are used to signal where the valence electrons are located around each cantlet in a Lewis structure. Virtually structures—specially those containing 2nd row elements—obey the octet rule, in which every cantlet (except H) is surrounded by eight electrons. Exceptions to the octet dominion occur for odd-electron molecules (gratis radicals), electron-deficient molecules, and hypervalent molecules.
Chemistry End of Affiliate Exercises
- Write the Lewis symbols for each of the post-obit ions:
(a) As3–
(b) I–
(c) Be2+
(d) O2–
(east) Gaiii+
(f) Li+
(g) N3–
- Many monatomic ions are found in seawater, including the ions formed from the post-obit listing of elements. Write the Lewis symbols for the monatomic ions formed from the following elements:
(a) Cl
(b) Na
(c) Mg
(d) Ca
(e) One thousand
(f) Br
(1000) Sr
(h) F
- Write the Lewis symbols of the ions in each of the following ionic compounds and the Lewis symbols of the atom from which they are formed:
(a) MgS
(b) Al2O3
(c) GaClthree
(d) K2O
(due east) Li3N
(f) KF
- In the Lewis structures listed here, M and X represent diverse elements in the 3rd flow of the periodic table. Write the formula of each chemical compound using the chemical symbols of each element:
(a)
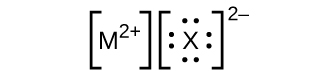
(b)
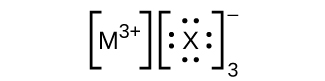
(c)
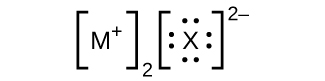
(d)
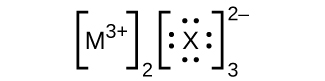
- Write the Lewis structure for the diatomic molecule P2, an unstable course of phosphorus plant in high-temperature phosphorus vapor.
- Write Lewis structures for the following:
(a) Htwo
(b) HBr
(c) PClthree
(d) SFtwo
(e) H2CCH2
(f) HNNH
(g) H2CNH
(h) NO–
(i) Nii
(j) CO
(yard) CN–
- Write Lewis structures for the following:
(a) O2
(b) H2CO
(c) AsF3
(d) ClNO
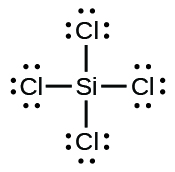
(e) SiCl4
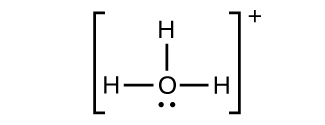
(f) HiiiO+
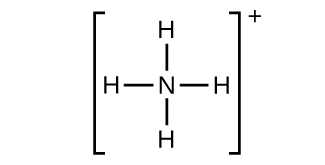
(g) NH4 +
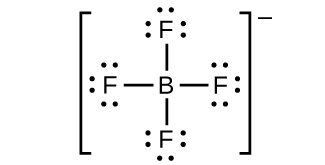
(h) BFfour −
(i) HCCH
(j) ClCN
(k) Cii 2+
- Write Lewis structures for the following:
(a) ClF3
(b) PCl5
(c) BF3
(d) PF6 −
- Write Lewis structures for the following:
(a) SeFsix
(b) XeF4
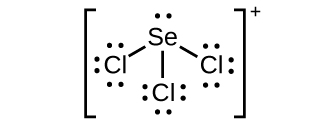
(c) SeCl3 +
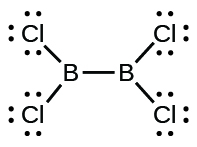
(d) Cl2BBCl2 (contains a B–B bond)
- Write Lewis structures for:
(a) PO4 3−
(b) ICliv −
(c) SO3 two−
(d) HONO
- Right the following statement: "The bonds in solid PbCltwo are ionic; the bond in a HCl molecule is covalent. Thus, all of the valence electrons in PbCl2 are located on the Cl– ions, and all of the valence electrons in a HCl molecule are shared between the H and Cl atoms."
- Write Lewis structures for the following molecules or ions:
(a) SbHiii
(b) XeF2
(c) Se8 (a cyclic molecule with a ring of viii Se atoms)
- Methanol, H3COH, is used every bit the fuel in some race cars. Ethanol, C2H5OH, is used extensively as motor fuel in Brazil. Both methanol and ethanol produce CO2 and HtwoO when they burn down. Write the chemical equations for these combustion reactions using Lewis structures instead of chemic formulas.
- Many planets in our solar system contain organic chemicals including marsh gas (CHfour) and traces of ethylene (C2H4), ethane (CiiHsix), propyne (HiiiCCCH), and diacetylene (HCCCCH). Write the Lewis structures for each of these molecules.
- Carbon tetrachloride was formerly used in fire extinguishers for electric fires. Information technology is no longer used for this purpose because of the formation of the toxic gas phosgene, Cl2CO. Write the Lewis structures for carbon tetrachloride and phosgene.
- Identify the atoms that correspond to each of the following electron configurations. So, write the Lewis symbol for the common ion formed from each atom:
(a) 1due south 22s ii2p v
(b) 1s ii2south 2twop 63s 2
(c) onesouthward 2twos twotwop 6threes 23p six4south 2threed ten
(d) onesouthward twoiisouth 22p six3south 23p six4s two3d ten4p 4
(due east) 1s two2s ii2p sixiiidue south iithreep 6ivs 23d 10ivp 1
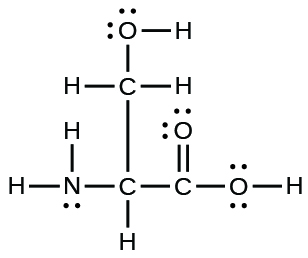
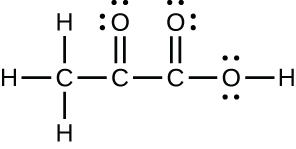
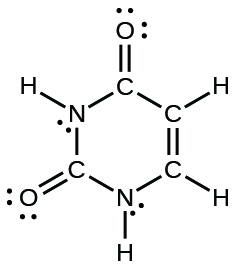
- The arrangement of atoms in several biologically important molecules is given here. Complete the Lewis structures of these molecules past adding multiple bonds and alone pairs. Do non add any more than atoms.
(a) the amino acrid serine:
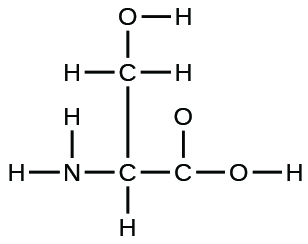
(b) urea:
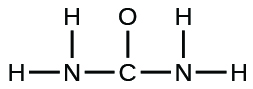
(c) pyruvic acid:
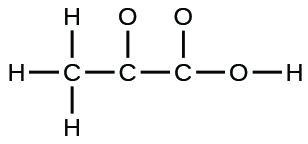
(d) uracil:
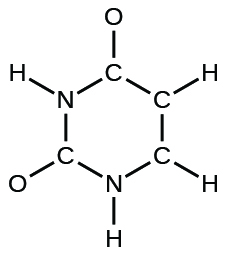
(e) carbonic acid:
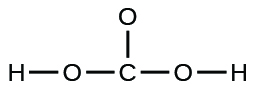
- A compound with a molar mass of about 28 chiliad/mol contains 85.7% carbon and 14.3% hydrogen by mass. Write the Lewis construction for a molecule of the compound.
- A compound with a molar mass of well-nigh 42 1000/mol contains 85.seven% carbon and 14.three% hydrogen by mass. Write the Lewis construction for a molecule of the chemical compound.
- Two arrangements of atoms are possible for a compound with a molar mass of nearly 45 thousand/mol that contains 52.2% C, 13.i% H, and 34.7% O by mass. Write the Lewis structures for the two molecules.
- How are single, double, and triple bonds similar? How do they differ?
Glossary
- double bond
- covalent bond in which two pairs of electrons are shared between 2 atoms
- gratuitous radical
- molecule that contains an odd number of electrons
- hypervalent molecule
- molecule containing at least one principal group element that has more than than eight electrons in its valence beat
- Lewis structure
- diagram showing lone pairs and bonding pairs of electrons in a molecule or an ion
- Lewis symbol
- symbol for an element or monatomic ion that uses a dot to stand for each valence electron in the element or ion
- lone pair
- two (a pair of) valence electrons that are not used to form a covalent bail
- octet rule
- guideline that states main group atoms will form structures in which viii valence electrons collaborate with each nucleus, counting bonding electrons as interacting with both atoms connected by the bond
- single bond
- bond in which a unmarried pair of electrons is shared between two atoms
- triple bail
- bond in which iii pairs of electrons are shared between ii atoms
Solutions
Answers to Chemistry Cease of Chapter Exercises
i. (a) eight electrons:
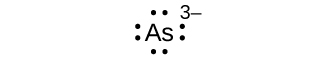 ;
;
(b) 8 electrons:
 ;
;
(c) no electrons
Be2+;
(d) eight electrons:
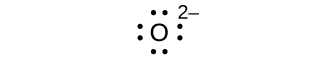 ;
;
(e) no electrons
Gathree+;
(f) no electrons
Li+;
(m) eight electrons:
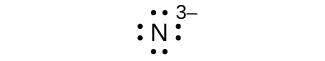
three. (a)
 ;
;
(b)
 ;
;
(c)
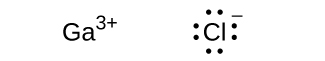 ;
;
(d)
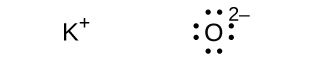 >;
>;
(e)
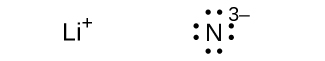 ;
;
(f)
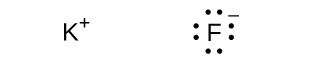
5.

seven. (a)
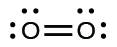
In this case, the Lewis structure is inadequate to depict the fact that experimental studies take shown 2 unpaired electrons in each oxygen molecule.
(b)
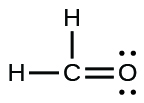 ;
;
(c)
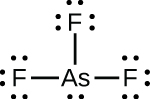 ;
;
(d)
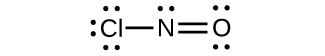 ;
;
(e)
 ;
;
(f)
 ;
;
(g)
 ;
;
(h)
 ;
;
(i)
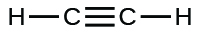 ;
;
(j)
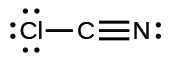 ;
;
(one thousand)
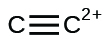
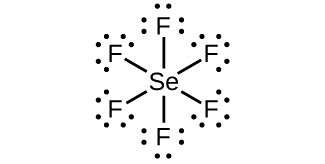
9. (a) SeFhalf-dozen:
 ;
;
(b) XeF4:
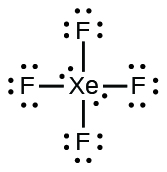 ;
;
(c) SeCl3 +:
 ;
;
(d) Cl2BBCl2:
11. Ii valence electrons per Lead atom are transferred to Cl atoms; the resulting Pb2+ ion has a half dozens 2 valence shell configuration. Two of the valence electrons in the HCl molecule are shared, and the other six are located on the Cl atom equally lone pairs of electrons.
13.

15.
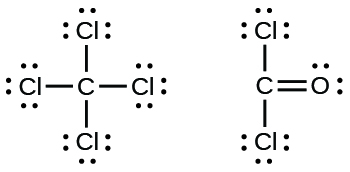
17. (a)
 ;
;
(b)
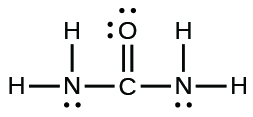 ;
;
(c)
 ;
;
(d)
 ;
;
(e)
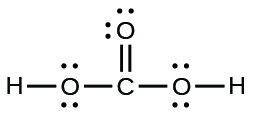
19.
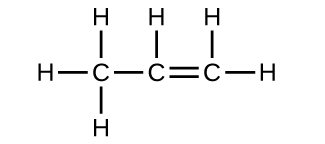
21. Each bond includes a sharing of electrons between atoms. Ii electrons are shared in a unmarried bond; iv electrons are shared in a double bond; and vi electrons are shared in a triple bond.
Source: https://opentextbc.ca/chemistry/chapter/7-3-lewis-symbols-and-structures/
Posted by: mcculloughglelavold.blogspot.com


0 Response to "Draw The Best Lewis Structure For Ccl+13. What Is The Formal Charge On The C?"
Post a Comment